A predictive journey towards trans-thioamides/amides†
Abstract
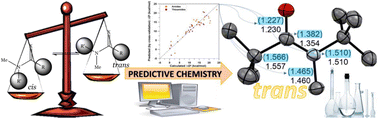
The cis–trans isomerization of (thio)amides was studied by DFT calculations to get the model for the higher preference for the cis conformation by guided predictive chemistry, suggesting how to select the alkyl/aryl substituents on the C/N atoms that lead to the trans isomer. Multilinear analysis, together with cross-validation analysis, helped to select the best fitting parameters to achieve the energy barriers of the cis to trans interconversion, as well as the relative stability between both isomers. Double experimental check led to the synthesis of the best trans candidate with sterically demanding t-butyl substituents, confirming the utility of predictive chemistry, bridging organic and computational chemistry.


 Please wait while we load your content...
Please wait while we load your content...