Metal-free hypervalent iodine-promoted tandem carbonyl migration and unactivated C(Ph)–C(Alkyl) bond cleavage for quinolone scaffold synthesis†
Abstract
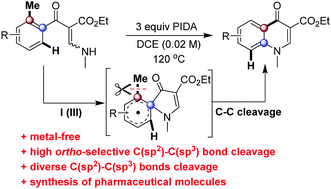
An unexpected iodine(III)-mediated C(sp3)–C(sp2) bond cleavage of 3-(methylamino)-2-(2-substitutedbenzoyl)acrylates for efficient synthesis of privileged scaffold 4-quinolones was described. Notably, a wide range of alkyl groups (e.g. methyl, tert-butyl or alkyl chain) can be conveniently cleaved in this system. The detailed mechanism studies revealed that the transformation proceeded through cascade ipso-cyclization and 1,2-carbonyl migration, the smaller bond energy determined ortho C–C bond cleavage rather than C–H bond cleavage, via an enamine radical intermediate.


 Please wait while we load your content...
Please wait while we load your content...