Na superionic conductor-type LiZr2(PO4)3 as a promising solid electrolyte for use in all-solid-state Li metal batteries
Abstract
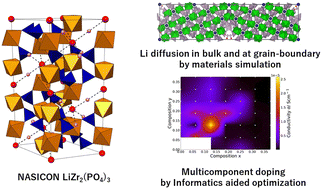
All-solid-state Li-ion batteries are of considerable interest as safer alternatives to Li-ion batteries containing flammable organic electrolytes. To date, however, achieving sufficient charging and discharging rates, in addition to capacity, at room temperature using these all-solid-state batteries has been challenging. To overcome these issues, material simulations and informatics investigations of a relatively new Na superionic conductor (NASICON)-type LiZr2(PO4)3 (LZP) electrolyte were conducted to elucidate its characteristics and material functions. The following thermodynamic and/or kinetic properties of NASICON-type Li-ion conductive oxides were investigated with respect to the crystal structure mainly using material simulation and informatics approaches: (1) the electrochemical stabilities of LZP materials with respect to Li metal and (2) Li-ion conductivities in the bulk and at the grain boundaries. An efficient materials informatics search method was employed to optimise the material functions of the LZP electrolyte via Bayesian optimisation. This study should promote the application of LZP in all-solid-state batteries for use in technologies such as mobile devices and electric vehicles and enable more complex composition and process control.


 Please wait while we load your content...
Please wait while we load your content...