Regulating force-resistance and acid-responsiveness of pure organics with persistent phosphorescence via simple isomerization†
Abstract
Stimulus-responsive purely organic room-temperature phosphorescence materials have been drawing massive attention due to their wide applications. Pyridine rings are introduced to supply π orbitals and cyanogroups are incorporated to boost the ISC efficiency by promoting the spin-forbidden transition. These groups are anticipated to enable the target molecule with multi-responsiveness because of the protonation of pyridine and their good crystallinity, which are able to regulate the acid-responsiveness and force-responsiveness, respectively. Based on the above design concept, four new D–A–A′ type molecules using carbazole as the donor and the pyridine ring and cyanogroup as acceptors were designed and synthesized. The D–A–A′ structure bestows these isomers with an evident intramolecular charge transfer (ICT) feature, particularly for 2-CNPyCZ and 3-CNPyCZ. All the isomers show intense long-lived phosphorescence with a lifetime over 500 ms. Particularly, 4-CNPyCZ has a high phosphorescence quantum yield of 27.1% owing to the strong intermolecular interactions that stabilize the 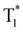 excitons. Interestingly, four isomers could retain their long-lived afterglow even after being heavily ground and the afterglow shows well resistance to external forces due to high crystallinity. 4-CNPyCZ manifests unique mechanochromism owing to the fluorescence shift and intensity change of phosphorescence. Moreover, the four isomers demonstrate distinctive acid-responsiveness and give out colorful emissions because the electron cloud dispersion of the nitrogen atom in the pyridine ring varied when altering the position of the cyanogroup. To the best of our knowledge, this is a limited work on room temperature phosphorescence about systematically regulating the responsiveness to external stimuli and proposing an effective molecular design strategy.
excitons. Interestingly, four isomers could retain their long-lived afterglow even after being heavily ground and the afterglow shows well resistance to external forces due to high crystallinity. 4-CNPyCZ manifests unique mechanochromism owing to the fluorescence shift and intensity change of phosphorescence. Moreover, the four isomers demonstrate distinctive acid-responsiveness and give out colorful emissions because the electron cloud dispersion of the nitrogen atom in the pyridine ring varied when altering the position of the cyanogroup. To the best of our knowledge, this is a limited work on room temperature phosphorescence about systematically regulating the responsiveness to external stimuli and proposing an effective molecular design strategy.



 Please wait while we load your content...
Please wait while we load your content...