Novel graphitic carbon nitride g-C9N10 as a promising platform to design efficient photocatalysts for dinitrogen reduction to ammonia: the first-principles investigation†
Abstract
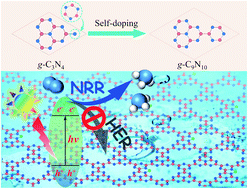
Photocatalysts play a vital role in the process of harnessing and converting solar energy into storable and valuable chemical fuels. As a polymeric semiconductor, graphitic carbon nitride g-C3N4 has been regarded as a next-generation photocatalyst due to its appealing band structure and excellent physicochemical stability. However, low electron conductivity and weak visible light absorption hinder its practical application. In this work, for the first time, we rationally designed a new graphitic carbon nitride, g-C9N10, based on a self-doping strategy by substituting the C3N3 ring for bridging the N atom in g-C3N4. g-C9N10 exhibits higher electron conductivity and longer wavelength absorption than those of g-C3N4, which make it a highly photoactive photocatalyst. Moreover, on the basis of “acceptance and donation” mechanism, single boron atom-decorated g-C9N10 (B@g-C9N10) can act as an efficient metal-free photocatalyst for nitrogen reduction reactions (NRRs) with high faradaic efficiency. Our results demonstrate that B@g-C9N10 can effectively activate and reduce N2 to NH3 with a low overpotential of 0.22 V, while the competitive hydrogen evolution reaction (HER) is efficiently suppressed. In addition, the photo-generated external potential is adequate to drive the NRR, endowing B@g-C9N10 with spontaneous NRR capability under visible/infrared light irradiation. This work may provide a new strategy for designing photocatalysts with high photoactivity and proposes g-C9N10 as a promising platform for designing efficient photocatalysts, which is expected to motivate more research efforts to explore.


 Please wait while we load your content...
Please wait while we load your content...