Uniform and dendrite-free zinc deposition enabled by in situ formed AgZn3 for the zinc metal anode†
Abstract
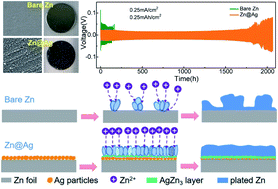
Rechargeable aqueous zinc-ion batteries (ZIBs) are attractive candidates for next-generation batteries. However, the challenge of uneven zinc electroplating/electrostripping on bare Zn anodes has severely restrained the practical application of ZIBs. To address this problem, a straightforward strategy of sliver particle-coated zinc plate (designated as Zn@Ag) via the metallic replacement reaction as the zinc metal anode has been employed to facilitate uniform and stable Zn-plating/stripping. The newly formed AgZn3 alloy from silver particles at the first zinc-plating cycle with good zinc affinity can effectively lower the energy barrier of zinc nucleation and promote uniform electric field distribution for the flow of Zn ions, resulting in stable zinc deposition. Benefiting from the in situ formed alloy phase, the Zn@Ag anode achieves stable cycling for over 1700 h with a very low polarization voltage of about 21 mV at 0.25 mA cm−2 and 0.25 mA h cm−2, while the bare Zn anode just exhibits less than 150 cycles with large voltage fluctuation and polarization under same conditions. As a consequence, greatly improved performance of the Zn@Ag//CNT/MnO2 full-cell with twice the capacity (177 mA h g−1) than that of bare Zn//CNT/MnO2 full-cell (71 mA h g−1) after 400 cycles at 0.6 A g−1 can be realized. This work provides a facile and effective approach to regulate Zn deposition for the achievement of long-life rechargeable ZIBs.


 Please wait while we load your content...
Please wait while we load your content...