Bipolar membrane polarization behavior with systematically varied interfacial areas in the junction region†
Abstract
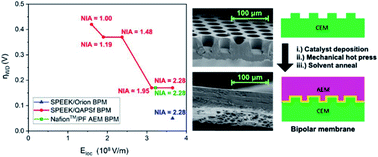
The palette of applications for bipolar membranes (BPMs) has expanded recently beyond electrodialysis as they are now being considered for fuel cell and electrolysis applications. Their deployment in emerging electrochemical technologies arises from the need to have a membrane separator that provides disparate pH environments and to prevent species crossover. Most materials research for BPMs has focused on water dissociation catalysts and less emphasis has been given to the design of the polycation–polyanion interface for improving BPM performance. Here, soft lithography fabricated a series of micropatterned BPMs with precise control over the interfacial area in the bipolar junction. Polarization experiments showed that a 2.28× increase in interfacial area led to a 250 mV reduction in the onset potential. Additionally, the same increase in interfacial area yielded marginal improvements in current density due to the junction region being under kinetics-diffusion control. A simple physics model based on the electric field of the junction region rationalized the reduction in the overpotential for water dissociation as a function of interfacial area. Finally, the soft lithography approach was also conducive for fabricating BPMs with different chemistries ranging from perfluorinated polymer backbones to alkaline stable poly(arylene) hydrocarbon polymers. These polymer chemistries are better suited for fuel cell and electrolysis applications. The BPM featuring the alkaline stable poly(terphenyl) anion exchange membrane had an onset potential of 0.84 V, which was near the thermodynamic limit, and was about 150 mV lower than a commercially available variant.


 Please wait while we load your content...
Please wait while we load your content...
