The impact of hydrogen valence on its bonding and transport in molten fluoride salts†
Abstract
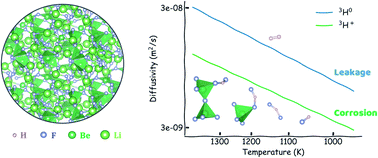
Interest in molten salts has increased significantly over the last decade due to their potential application in various clean-energy technologies including hydrogen generation, solar heat storage, advanced fission nuclear power plants, and compact fusion energy systems. In nuclear fission and fusion power plants, high heat capacity molten salts allow operation at high temperature and atmospheric pressure, which could dramatically increase efficiency, reduce capital cost, and enable passive safety features. In many of these systems, the hydrogen isotope is of particular importance due to its ability to corrode structural materials as 3H+ in fluoride salts, and its potential to cause significant radioactive release as diffusive 3H0, which are cited as key barriers to technological deployment. Yet, the chemistry and transport behavior of the hydrogen species remain poorly understood due to the difficulties in handling toxic salts and radioactive materials. Here, using ab initio molecular dynamics, we present a coupled examination of hydrogen speciation in the most common prototypical salts 66.6% LiF–33.3% BeF2 (Flibe) and 46.5% LiF–11.5% NaF–42% KF (Flinak). Using extensively validated calculations on the local structure and dynamics, we find significant difference between 3H0 and 3H+ transport behaviors that are usually overlooked. We find that 3H0, which always exists as H2, diffuses 3–5 times faster than 3H+, which can be ascribed to hydrogen bonding and complexation in solution. This work explains contradicting experimental results and provides useful species transport data for designing hydrogen capture and corrosion control systems for molten salts.


 Please wait while we load your content...
Please wait while we load your content...