Construction of a hydroxide responsive C3-symmetric supramolecular gel for controlled release of small molecules†
Abstract
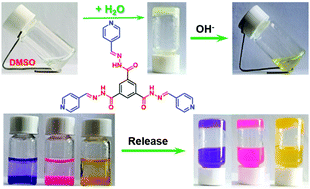
A C3-symmetric acylhydrazone-based low molecular weight gelator (BHTP) bearing three pyridine units was synthesized and it was found to form a stable supramolecular gel in the mixture solvent of DMSO–H2O. The morphology of the gel as observed by FE-SEM showed a dense sheet structure. Hydrogen bonding and π–π stacking between the gelators were determined as the non-covalent interactions for the gelation, which were investigated thoroughly using XRD, UV-Vis, 1H NMR and FT-IR instruments. BHTP could form pH tolerant supramolecular gels in the widest range of pH values from 1 to 11. The DMSO–H2O (v : v = 1 : 1) gel exhibited selective response to OH− over a series of other anions through the color change from a white gel to a yellow solution, and the OH− response mechanism was proved by 1H NMR experiments. In solution, the lowest detection limit of BHTP for OH− was calculated to be as low as 1.62 × 10−7 M via UV-Vis titration experiments. Finally, encapsulation and controlled release of small molecules such as rhodamine B, crystal violet and methyl orange have been successfully carried out, demonstrating the potential for drug delivery application of this C3-symmetric supramolecular gel. This work opens a novel avenue for the preparation of supramolecular gel-based multiple functional smart materials.


 Please wait while we load your content...
Please wait while we load your content...