Chain conformation of polymers adsorbed to clay particles: effects of charge and concentration†
Abstract
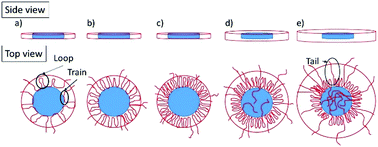
High molecular weight polymers are used in industrially important applications where the polymers adsorb onto surfaces of inorganic particles and facilitate dispersion, stabilization or flocculation of formulations. The surface conformation of these adsorbed polymers is key to their performance, yet an understanding of the effects of polymer charge, concentration and molecular weight on polymer conformation is incomplete. Here the adsorption behavior of high molecular weight, non-ionic, polyethylene oxide (PEO) and anionic partially hydrolyzed polyacrylamide (HPAM) polymers to a model clay, LAPONITE® (a trademark of BYK Additives, Inc.), was studied by small angle neutron scattering over a range of polymer concentrations. The adsorption of PEO was dependent on polymer concentration at all molecular weights, first adsorbing onto the radial edge of the clay particles and then to the clay faces as polymer concentration increased. While similar behavior was observed at low concentrations of HPAM, above a critical concentration, HPAM desorption results in large clay aggregates, which decrease in size at higher polymer loadings. The difference in adsorption properties as a function of polymer charge and concentration is interpreted in terms of conformational differences of adsorbed polymer at the clay surface. Changes in the volume fraction of adsorbed PEO with polymer concentration indicate changes in the relative amounts of adsorbed chain segments in trains, loops and tails, with no clay aggregation. In contrast, a constant volume fraction of adsorbed HPAM at low concentrations gives way to desorption at higher concentration due to charge repulsion, and depletion aggregation of the clay.


 Please wait while we load your content...
Please wait while we load your content...