Nickel-catalyzed enantioselective 1,2-vinylboration of styrenes†
Abstract
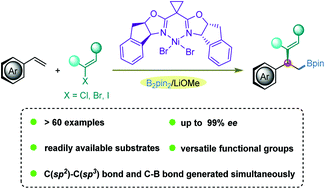
A novel nickel-catalyzed asymmetric 1,2-vinylboration reaction has been developed to afford benzylic alkenylboration products with high yields and excellent enantioselectivities by using a chiral bisoxazoline ligand. Under optimized conditions, a wide variety of chiral 2-boryl-1,1-arylvinylalkanes are efficiently prepared from readily available olefins and vinyl halides in the presence of bis(pinacolato)diboron as the boron source in a mild and easy-to-operate manner. This three-component cascade protocol furnishes exceptional chemo- and stereoselectivity, and its usefulness is illustrated by its application in asymmetric modifications of several structurally complex natural products and pharmaceuticals.


 Please wait while we load your content...
Please wait while we load your content...