Solvothermal synthesis of poly(acrylic acid) decorated magnetic molybdenum disulfide nanosheets for highly-efficient adsorption of cationic dyes from aqueous solutions†
Abstract
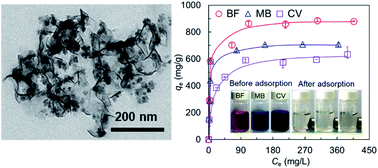
Herein we report solvothermal synthesis of poly(acrylic acid) (PAA) decorated magnetic molybdenum disulfide nanosheets (MMoS2/PAA) for highly efficient adsorption of three cationic dyes of basic fuchsin (BF), methylene blue (MB), and crystal violet (CV) from aqueous solutions. The synthesized MMoS2/PAA was characterized by several techniques including transmission electron microscopy (TEM), Fourier transform-infrared spectroscopy (FT-IR), X-ray photoelectron spectroscopy (XPS), thermogravimetric analysis (TGA), vibrating sample magnetometry (VSM), and dynamic light scattering (DLS). Due to the strong electrostatic interaction between cationic dyes and the anionic nanosheet surface, the obtained MMoS2/PAA showed ultrafast adsorption of BF, MB and CV within 2 min with high adsorption capacities of 886.1, 709.0, and 633.6 mg g−1, respectively, much higher than those materials reported recently. PAA molecules bound on the nanosheets played a crucial role in significantly enhancing the dye adsorption. The adsorption kinetics and isotherms of three dyes onto the MMoS2/PAA were well described by the pseudo-second-order kinetic and the Langmuir models. Moreover, the MMoS2/PAA also exhibited high removal efficiencies for various mixed-dye solutions. Besides, such a functional nanomaterial could be effectively recovered from dye solutions under an external magnetic field and reused for dye adsorption without compromising on its performance indicating it can serve as an excellent adsorbent for effective removal of a variety of cationic organic pollutants from industrial effluents.


 Please wait while we load your content...
Please wait while we load your content...