Understanding liquefaction in halide perovskites upon methylamine gas exposure†
Abstract
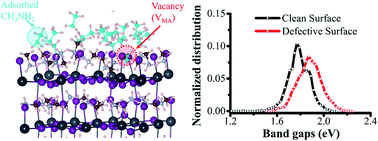
Methylamine (CH3NH2, MA) gas-induced fabrication of organometal CH3NH3PbI3 based perovskite thin films are promising photovoltaic materials that transform the energy from absorbed sunlight into electrical power. Unfortunately, the low stability of the perovskites poses a serious hindrance for further development, compared to conventional inorganic materials. The solid-state perovskites are liquefied and recrystallized from CH3NH2. However, the mechanism of this phase transformation is far from clear. Employing first principles calculations and ab initio molecular dynamics simulations, we investigated the formation energy of primary defects in perovskites and the liquefaction process in CH3NH2 vapor. The results indicated that defect-assisted surface dissolution leads to the liquefaction of perovskite thin films in CH3NH2 vapor. Two primary defects were studied: one is the Frenkel pair defect (including both negatively charged interstitial iodide ion (Ii−) and iodide vacancy (VI+) at the PbI2-termination surface, and the other is the Schottky defects (methylammonium vacancy, VMA) at the MAI-termination surface. Moreover, the defect-induced disorder in the microstructure reduces the degeneration of energy levels, which leads to a blue shift and broader absorption band gap, as compared to the clean perovskite surface. The mechanism of how defects impact the surface dissolution could be applied for the further design of high-stability perovskite solar cells.


 Please wait while we load your content...
Please wait while we load your content...