Highly sensitive and selective colorimetric detection of dual metal ions (Hg2+ and Sn2+) in water: an eco-friendly approach†
Abstract
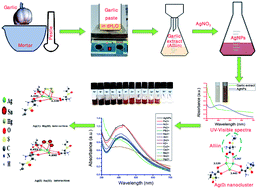
Application of an alliin-based precursor for the synthesis of silver nanoparticles (Ag NPs) which is an emerging, reliable and rapid sensor of heavy metal ion contaminants in water is reported here. The Ag NPs were characterized by using UV-visible spectroscopy, X-ray diffraction, transmission electron microscopy, X-ray photoelectron spectroscopy and Raman spectroscopy analysis techniques. The Ag NPs simultaneously and selectively detect Hg2+ and Sn2+ ions from aqueous solution. The sensitivity and selectivity of the prepared Ag NPs towards other representative transition-metal ions, alkali metal ions and alkaline earth metal ions were also studied. For more precise evidence, a density functional theory study was carried out to understand the possible mechanism and interaction in the detection of Hg2+ and Sn2+ by Ag NPs. The limits of detection for Hg2+ and Sn2+ ions were found as 15.7 nM and 11.25 nM, respectively. This assay indicates the possible use of garlic extract-synthesized Ag NPs for sensing Hg2+ and Sn2+ in aqueous solution very significantly. So, the simple, green, eco-friendly and easy method to detect the dual metal ions may further lead to a potential sensor of heavy metal ion contaminants in water of industrial importance.


 Please wait while we load your content...
Please wait while we load your content...