Synthesis and characterization of novel protein nanodots as drug delivery carriers with an enhanced biological efficacy of melatonin in breast cancer cells†
Abstract
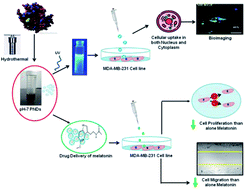
Melatonin is a potent antioxidant, chemotherapeutic and chemo preventive agent against breast cancer. However, its short half-life is one of the major limitations in its application as a therapeutic drug. To overcome this issue, the green-emitting protein nanodot (PND) was synthesized by a one-step hydrothermal method for loading melatonin. The synthesized pH-7 and pH-2 PND showed a quantum yield of 22.1% and 14.0%, respectively. The physicochemical characterization of both PNDs showed similar morphological and functional activities. Furthermore, the biological efficacy of melatonin-loaded PND (MPND) was evaluated in a breast cancer cell line (MDA-MB-231) for live-cell imaging and enhanced nano-drug delivery efficacy. Interestingly, the permeability of neutral pH PND in both cell cytoplasm and nucleus nullifies the limitations of real-time live-cell imaging, and ensures nuclear drug delivery efficacy. Neutral pH PND showed better cell viability and cytotoxicity as a fluorescence bioimaging probe compared to acidic PND. The bioavailability and cell cytotoxicity effect of MPND on MDA-MB-231 breast cancer cells were studied through confocal and migration assay. Results showed that MPND causes enhanced bioavailability, better cellular uptake, and inhibition of the migration of breast cancer cells as compared to the drug alone. Besides, the synthesized MPND showed no sign of fluorescence quenching even at a high concentration of melatonin, making it an ideal nanocarrier for bioimaging and drug delivery.


 Please wait while we load your content...
Please wait while we load your content...