Cocatalyst versus precatalyst impact on the vinyl-addition polymerization of norbornenes with polar groups: looking at the other side of the coin†
Abstract
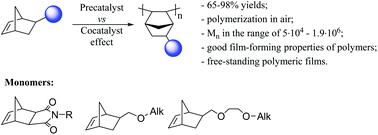
Polymers derived from norbornenes with polar groups are of interest as modular templates and materials for membrane processes and microelectronic applications due to the attractive combination of their properties. However, the synthesis of high-molecular weight homopolymers from norbornenes containing polar groups is still a challenge. Herein, we report our examination of the precatalyst and cocatalyst influence on vinyl-addition polymerization of two types of norbornene monomers with bulky and polar groups (substituted nadimides and norbornenes with cellosolve motif). The key to successful vinyl addition polymerization of these monomers is the proper combination of a cocatalyst and a Pd precatalyst. Both components of a catalytic system seem to play equal roles. The catalytic activity and molecular weights of the resulting polymers are efficiently tuned by the nature of both pre- and cocatalyst. Moreover, the impact of the cocatalyst depends on the monomer nature and can be different for norbornenes with nonpolar and polar groups. As a result, a set of glassy and amorphous vinyl-addition homopolymers from norbornenes with polar groups is prepared with yields up to 98% and Mw ≤ 3 × 106 over Pd complexes with N-heterocyclic carbene ligands. The polymers exhibit excellent film-forming properties. These results provide an insight and opportunity for further work expansion in order to obtain new high-performance polymers based on strained cycloalkenes with polar groups, which are not currently available.


 Please wait while we load your content...
Please wait while we load your content...