Biobased poly(ester-co-glycoside) from reactive natural Brønsted acidic deep eutectic solvent analogue†
Abstract
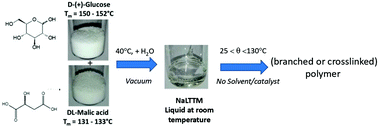
In the framework of green chemistry applied to polymeric-based materials, (re-)designing synthetic methodologies and processes that use and generate substances with low(er) impact to human and environment has been the topic of intense research, considering in particular the strategic place occupied by solvents. In this context, the aim of this work was to evaluate the possibility of synthesizing carbohydrate polymers from Natural Brønsted Acidic Low Transition Temperature Mixtures, including glucose and malic acid in a 1 : 1 ratio as monomers, without the use of any solvent or catalyst. Mixtures that are liquid at room temperatures were obtained by evaporation of aqueous mixtures of glucose and malic acid, which associated to their hygroscopicity, makes it more relevant to consider {glucose : malic acid : water} (1 : 1 : x molar) ternary rather than binary mixtures. The impact of water content on the properties of the mixtures before reaction is first discussed. Then polymerization is presented: as a function of the temperature, but still lower than the melting temperature of the starting monomers, not only ester but also glycosidic bonds are revealed, such that poly(ester-co-glycoside)s with various ester content result. Moreover, by post-curing the polymers for longer times, a crosslinked thermoset polymer with high gel content and crosslink density is obtained, as demonstrated by dye release experiments.

 Please wait while we load your content...
Please wait while we load your content...