Synthesis and properties of l-lactide/1,3-dioxolane copolymers: preparation of polyesters with enhanced acid sensitivity†
Abstract
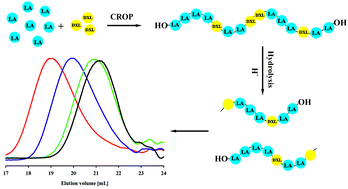
Herein, we report the first example of cationic ring-opening copolymerization of 5-membered cyclic acetal (1,3-dioxolane (DXL)) with L-lactide (LA) to afford polylactide containing acetal units. Copolymers with different compositions (containing up to 27 mol% DXL) were obtained in the presence of 1,2-ethanediol as an initiator and triflic acid as a catalyst. The incorporation of DXL into the polyester chain was examined by 1H and 13C NMR. Additionally, two-dimensional nuclear magnetic resonance spectroscopy (heteronuclear single-quantum correlation spectroscopy (HSQC)) was used for the identification of DXL units in the polyester chain. The obtained copolyesters were amorphous and exhibited glass transition temperatures (Tg) that were strongly dependent on the copolymer composition and ranged from 8 °C to 32 °C. When subjected to hydrolysis in an acidic environment, PLA/PDXL copolymers showed accelerated degradation in comparison with the PLA homopolymer. The new polyesters containing acid-sensitive sequences may extend the field of degradable materials.


 Please wait while we load your content...
Please wait while we load your content...