Characterisation of a hyperthermophilic transketolase from Thermotoga maritima DSM3109 as a biocatalyst for 7-keto-octuronic acid synthesis†
Abstract
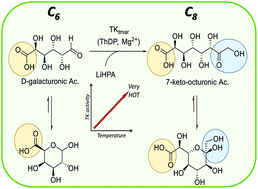
Transketolase (TK) is a fundamentally important enzyme in industrial biocatalysis which carries out a stereospecific carbon–carbon bond formation, and is widely used in the synthesis of prochiral ketones. This study describes the biochemical and molecular characterisation of a novel and unusual hyperthermophilic TK from Thermotoga maritima DSM3109 (TKtmar). TKtmar has a low protein sequence homology compared to the already described TKs, with key amino acid residues in the active site highly conserved. TKtmar has a very high optimum temperature (>90 °C) and shows pronounced stability at high temperature (e.g. t1/2 99 and 9.3 h at 50 and 80 °C, respectively) and in presence of organic solvents commonly used in industry (DMSO, acetonitrile and methanol). Substrate screening showed activity towards several monosaccharides and aliphatic aldehydes. In addition, for the first time, TK specificity towards uronic acids was achieved with TKtmar catalysing the efficient conversion of D-galacturonic acid and lithium hydroxypyruvate into 7-keto-octuronic acid, a very rare C8 uronic acid, in high yields (98%, 49 mM).
- This article is part of the themed collection: Biocatalysis: A cross-journal collection


 Please wait while we load your content...
Please wait while we load your content...