Facilitating room-temperature oxygen ion migration via Co–O bond activation in cobaltite films†
Abstract
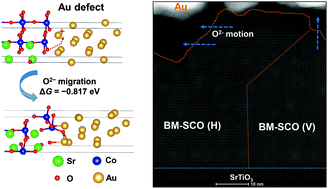
Oxygen ion migration in strongly correlated oxides can cause dramatic changes in the crystal structure, chemical and magnetoelectric properties, which holds promising for a wide variety of applications in catalysis, energy conversion, and electronics. However, the high strength and stability of metal–oxygen (M–O) bonds cause a large thermodynamic barrier for oxygen migration. Here, we designed Co–O bond activation in cobaltite (SrCoOx) films by Au-nanodot-decoration. Charge transfer from Au to SrCoOx effectively weakens the Co–O bond, meanwhile Co–O–Au synergistic bonding remarkably decreases the migration barrier of oxygen ions. Fast oxygen evolution occurs at the perimeter of the Au/SrCoOx interface, and the chemical potential gradient of O2− drives inner ion diffusion to the surface. Consequently, bias-free topotactic phase reduction from perovskite SrCoO3−δ to brownmillerite SrCoO2.5 has been achieved at room temperature. Our finding explores a new dimension to accelerate oxygen ion kinetics in transition-metal oxides from the aspect of interfacial bond activation, which is significant for developing oxide/noble-metal interfaces for high-efficiency ion migration and redox catalysis at low temperature.


 Please wait while we load your content...
Please wait while we load your content...