Photochemistry of triphenylamine (TPA) in homogeneous solution and the role of transient N-phenyl-4a,4b-dihydrocarbazole. A steady-state and time-resolved investigation†
Abstract
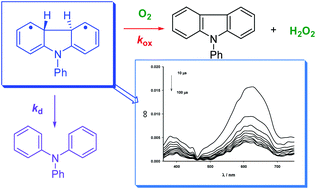
The photoreactivity of triphenylamine in homogeneous media has been investigated by means of laser flash photolysis spectroscopy and preparative experiments. The goal of this study consists in the evaluation of the photophysical and photochemical behavior of transient N-phenyl-4a,4b-dihydrocarbazole (DHC0) under polar and non-polar solvents which was not performed yet. The kinetic parameters of such a key intermediate including lifetime (τd, τR and τox) and rate constants (kd, kR and kox) have been measured and the intermediacy of DHC0 in the co-oxidation of nucleophilic substrates such as sulfides and phosphines was investigated by a combined time-resolved and steady-state approach. Two distinctly oxidizing intermediates, a hydroperoxy radical and a hydroperoxide derivative were formed from transient DHC0 in the presence of molecular oxygen. Radical I oxidizes triarylphosphines, whereas intermediate II converts sulfides into the corresponding sulfoxides.


 Please wait while we load your content...
Please wait while we load your content...