A series of asymmetric and symmetric porphyrin derivatives: one-pot synthesis, nonlinear optical and optical limiting properties†
Abstract
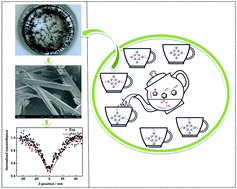
In this work, a series of asymmetric and symmetric porphyrin derivatives (structure types: A4, A3B1, trans-A2B2, cis-A2B2, A1B3, and B4) have been synthesized via a one-pot method and characterized by TLC, HPLC, FT-IR, 1H-NMR and UV-Vis to identify their structures and properties. The porphyrin derivatives with trans-A2B2 and cis-A2B2 structures are visually distinguished via single-crystal X-ray diffraction; the crystal morphology of trans-A2B2 is a needle-like structure. All porphyrin derivatives (I–VI) have excellent nonlinear optical (NLO) and optical limiting (OL) properties, and are investigated using the standard Z-scan technique. Porphyrin derivative III exhibits the maximum nonlinear absorption, nonlinear refraction coefficients and optical limitation threshold, and the values are 3.7 × 10−9 m W−1, −17 × 10−17 m2 W−1 and 0.11 J cm−2, respectively. The results demonstrate that the introduction of different substituent numbers (mono-, di- and tri-, tetra-) and substituent sites (trans-A2B2 and cis-A2B2) in the porphyrins could influence the NLO and OL properties. Structure regulation of porphyrin can lead to the adjustment of NLO and OL properties, and affect its practical NLO application in the future.


 Please wait while we load your content...
Please wait while we load your content...