A novel water-soluble multicolor halo- and photochromic switching system based on a nitrile-rich acceptor†
Abstract
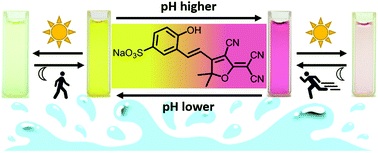
A directed synthesis of the first nitrile-rich dye showing visible-light-induced negative photochromism in aqueous medium was implemented. The possibility to change the color of an aqueous solution from yellow to magenta by varying the pH from 5 to 8 was shown. The synthesized dye was found to be a thermally unstable photochrome (T-type) whose photoinduced form returned to the initial state following the first order kinetic equation in the dark at room temperature. It was possible to change the dark reaction rate in a wide range and to block the photochromic process by pH regulation. The calculated pKa1 of the initial form was more than 2.5 units higher than the pKa1S of the photoinduced form, which indicated the photoacidity of the dye in aqueous medium. This allowed the pH of the solution on 1.2 pH units to be reversibly changed under visible light irradiation, followed by dark relaxation. The obtained results revealed a new direction in the investigation of nitrile-rich negative photochromes, namely, the synthesis of water-soluble multicolor molecular switching systems sensitive to several factors.


 Please wait while we load your content...
Please wait while we load your content...