Reductive desulfurization of aromatic sulfides with nickel boride in deep eutectic solvents†
Abstract
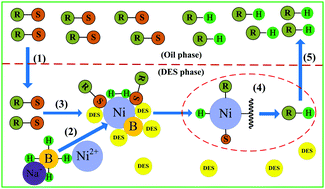
In order to improve the desulfurization rate, deep eutectic solvents (DESs) were used for the first time as solvents in the reductive desulfurization process with nickel boride (NB). DES plays a dual role of stabilizer and extracting agent in the desulfurization process. NBs prepared in different solvents were characterized by PSZPA, SAPA, XRD and FTIR spectroscopy. Results indicate that the NBs with small particle size and high specific surface area can be obtained by using DES as the solvent. The desulfurization rate of aromatic sulfides using DESs as the solvent is much higher than that using MeOH/THF as the solvent. The desulfurization rate in different DESs follows the order of tetrabutyl ammonium chloride (TBAC)/polyethylene glycol > TBAC/ethylene glycol > TBAC/glycerol. No chemical reactiong takes place between NB and DESs, and the structures of the DESs remain unchanged after regeneration.


 Please wait while we load your content...
Please wait while we load your content...