Label-free single-cell isolation enabled by microfluidic impact printing and real-time cellular recognition†
Abstract
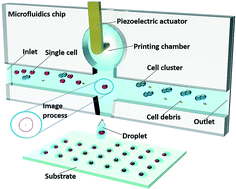
Analysis of cellular components at the single-cell level is important to reveal cellular heterogeneity. However, current technologies to isolate individual cells are either label-based or have low performance. Here, we present a novel technique by integrating real-time cellular recognition and microfluidic impact printing (MIP) to isolate single cells with high efficiency and high throughput in a label-free manner. Specifically, morphological characteristics of polystyrene beads and cells, computed by an efficient image processing algorithm, are utilized as selection criteria to identify target objects. Subsequently, each detected single-cell object in the suspension is ejected from the microfluidic channel by impact force. It has been demonstrated that the single-cell isolating system has the ability to encapsulate polystyrene beads in droplets with an efficiency of 95%, while for HeLa cells, this has been experimentally measured as 90.3%. Single-cell droplet arrays are generated at a throughput of 2 Hz and 96.6% of the cells remain alive after isolation. This technology has significant potential in various emerging applications, including single-cell omics, tissue engineering, and cell-line development.


 Please wait while we load your content...
Please wait while we load your content...