Facile and sustainable etherification of ethyl cellulose towards excellent UV blocking and fluorescence properties
Abstract
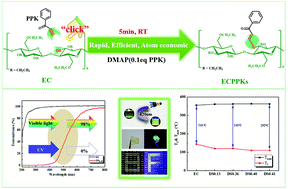
Chemical modification of cellulose to prepare functional cellulose derivatives could enable the utilization of cellulose in various promising applications. However, the conventional methods for cellulose functionalization suffer from severe drawbacks, especially the environmental aspects, due to the use of toxic reagents and the generation of large amounts of waste. Herein, a more sustainable way, the novel hydroxyl–yne click reaction, is proposed to synthesize cellulose derivatives. The hydroxyl groups of ethyl cellulose (EC) were reacted with the alkyne groups of 1-phenyl-2-propargyl-1-ketone (PPK) at room temperature for preparing new kinds of ethyl cellulose phenyl propylene ketone ether derivatives (ECPPKs). More than 80% substitution was rapidly completed within the first 5 min and 82% of the residual hydroxyl groups (–OHC3) in EC could be substituted, demonstrating the high efficiency of the hydroxyl–yne click reaction for the modification of cellulosic materials. FTIR and NMR results proved the successful grafting of PPK via a vinyl ether linkage (–C–O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–). The introduction of PPK moieties widened the temperature window for the melt processing of cellulose, which is beneficial for the thermo processing of cellulosic materials. Furthermore, owing to the existence of the newly formed vinyl ether linkage, ECPPKs showed almost 100% shielding ratio for UV light and visible light (420 nm) excited fluorescence properties. This facile and efficient method provides a more sustainable strategy for the functionalization of cellulosic materials, expanding its application in UV-blocking and fluorescent material fields.
C–). The introduction of PPK moieties widened the temperature window for the melt processing of cellulose, which is beneficial for the thermo processing of cellulosic materials. Furthermore, owing to the existence of the newly formed vinyl ether linkage, ECPPKs showed almost 100% shielding ratio for UV light and visible light (420 nm) excited fluorescence properties. This facile and efficient method provides a more sustainable strategy for the functionalization of cellulosic materials, expanding its application in UV-blocking and fluorescent material fields.


 Please wait while we load your content...
Please wait while we load your content...