Aryl acyl peroxides for visible-light induced decarboxylative arylation of quinoxalin-2(1H)-ones under additive-, metal catalyst-, and external photosensitizer-free and ambient conditions†
Abstract
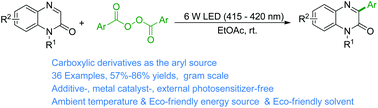
Aryl radicals were generated for the first time from cheap and easily available aryl acyl peroxides in eco-friendly ethyl acetate under ambient conditions and visible-light illumination in the absence of any additive, metal catalyst, or external photosensitizer. The present arylation of quinoxalin-2(1H)-ones was chemo- and regioselective, and provided good access to various 3-arylquinoxalin-2(1H)-ones.
- This article is part of the themed collection: 2021 Green Chemistry Hot Articles


 Please wait while we load your content...
Please wait while we load your content...