A GPx-mimetic copper vanadate nanozyme mediates the release of nitric oxide from S-nitrosothiols†
Abstract
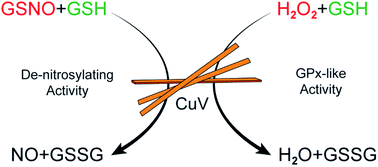
Although reactive oxygen and nitrogen species (ROS/RNS), such as hydrogen peroxide (H2O2), nitric oxide (NO), hydroxyl radicals (OH˙), superoxide (O2−) etc., play crucial roles in redox biology and cellular signaling, higher concentrations of these species lead to oxidative and nitrosative stress, which are associated with various pathophysiological conditions like neurodegeneration, cardiovascular diseases and cancer. There is growing evidence that functional impairment of the endothelium is one of the first recognizable signs of the development of atherosclerotic cardiovascular disease. A decreased bioavailability of NO and increased generation of ROS are the two major molecular changes associated with endothelial dysfunction. Therefore, it is a viable strategy to increase the bioavailability of NO while reducing the amount of ROS to prevent the progression of cardiovascular diseases. In this paper, we discuss for the first time that copper vanadate (CuV2O6) can not only release NO from S-nitrosothiols but can also control the ROS levels by functionally mimicking the antioxidant enzyme glutathione peroxidase (GPx) at physiological pH. We used several imaging techniques and spectroscopic measurements to understand the catalysis on the surface of the material during the reactions. The denitrosylation, as well as GPx-like activity, by CuV2O6 can be carried out multiple times without affecting the catalytic activity.
- This article is part of the themed collection: Natural and artificial metalloenzymes


 Please wait while we load your content...
Please wait while we load your content...