NMR and luminescence experiments reveal the structure and symmetry adaptation of a europium ionic liquid to solvent polarity†
Abstract
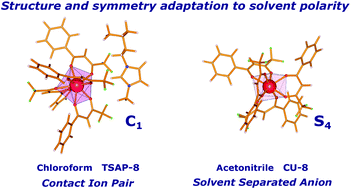
By combining NMR data (nuclear Overhauser effect and pseudocontact shifts) with luminescence measurements, we uncover how the structure of an anionic europium complex adapts to different solvent polarities as a result of the different relative proximities of the ion pairs. In nonpolar solvents, the detected contact ion pairs, CIPs, indicate that the ions remain in proximity, with the molecular cation, and then perturbing and distorting the coordination polyhedron of the anion complex to a low symmetry configuration, which promotes luminescence. Differently, solvent separated ion pairs occur in polar solvents, indicating that the molecular ions have been disconnected. Thus, in polar solvents, the europium complex anion becomes free from the close influence of the molecular cation, allowing the coordination polyhedron to increase its symmetry, which in turn reduces the luminescence of the anionic europium complex. This reduction of coordination polyhedron symmetry by the close proximity of the molecular cation in nonpolar solvents was confirmed by additional photophysical measurements combined with quantum chemical RM1 calculations, suggesting that, in nonpolar solvents, the symmetry point group of the coordination polyhedron is C1, whereas in polar solvents it is either D2d or S4. The nonpolar solvents used were benzene, chloroform and dichloromethane; and the polar ones were acetone and acetonitrile. The synthesized ionic liquids were tetrakis [C5mim][La(BTFA)4] and [C5mim][Eu(BTFA)4], where BTFA stands for 4,4,4-trifluoro-1-phenyl-1,3-butanedione, lanthanoids (La3+ and Eu3+) and C5mim stands for 1-methyl-3-isopentylimidazolium. They were synthesized by a microwave methodology that is both fast and green (the synthetic reaction takes about 15 min) and also leads to more easily purifiable crystals.


 Please wait while we load your content...
Please wait while we load your content...