Photorelease of a metal-binding pharmacophore from a Ru(ii) polypyridine complex†
Abstract
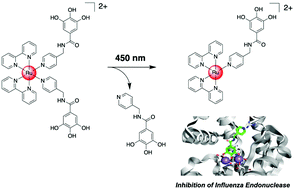
The adoption of compounds that target metalloenzymes comprises a relatively low (<5%) percentage of all FDA approved therapeutics. Metalloenzyme inhibitors typically coordinate to the active site metal ions and therefore contain ligands with charged or highly polar functional groups. While these groups may generate highly water-soluble compounds, this functionalization can also limit their pharmacological properties. To overcome this drawback, drug candidates can be formulated as prodrugs. While a variety of protecting groups have been developed, increasing efforts have been devoted towards the use of caging groups that can be removed upon exposure to light to provide spatial and temporal control over the treatment. Among these, the application of Ru(II) polypyridine complexes is receiving increased attention based on their attractive biological and photophysical properties. Herein, a conjugate consisting of a metalloenzyme inhibitor and a Ru(II) polypyridine complex as a photo-cage is presented. The conjugate was designed using density functional theory calculations and docking studies. The conjugate is stable in an aqueous solution, but irradiation of the complex with 450 nm light releases the inhibitor within several minutes. As a model system, the biochemical properties were investigated against the endonucleolytic active site of the influenza virus. While showing no inhibition in the dark in an in vitro assay, the conjugate generated inhibition upon light exposure at 450 nm, demonstrating the ability to liberate the metalloenzyme inhibitor. The presented inhibitor–Ru(II) polypyridine conjugate is an example of computationally-guided drug design for light-activated drug release and may help reveal new avenues for the prodrugging of metalloenzyme inhibitors.
- This article is part of the themed collection: The central role of the d-block metals in the periodic table


 Please wait while we load your content...
Please wait while we load your content...