Promotional effect of indium on Cu/SiO2 catalysts for the hydrogenation of dimethyl oxalate to ethylene glycol†
Abstract
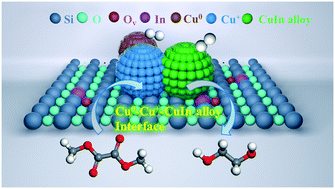
The synthesis of ethylene glycol (EG) through hydrogenation of syngas-derived dimethyl oxalate (DMO) has been a promising method because syngas can be obtained from plentiful resources such as coal, natural gas, biomass, etc. In this work, we fabricated several indium-promoted Cu/SiO2 catalysts by a one-pot hydrolysis precipitation (HP) method for the DMO-to-EG reaction. The textural and physiochemical properties of the catalysts were revealed using multiple characterization methods. The intimate contact of Cu and In enhances the reduction of indium oxide and the formation of CuIn alloy. The introduction of indium also markedly improves the copper dispersion and formation of Cu0 active sites, which improve the activation of H2. The plentiful interface of Cu+–CuIn alloy prompts the conversion of the carbonyl group adsorbed on the Cu+ sites with the dissociated hydrogen on the vicinal CuIn alloy, which is confirmed by the higher TOF (Cu+) and the lower apparent activation energy (Ea) on the Cu1In/SiO2 catalyst. Both CuIn alloy and Cu0 species have a synergistic effect with Cu+, endowing the Cu1In/SiO2 catalyst with a higher EG yield (96%) in comparison with the Cu/SiO2 catalyst without doping.


 Please wait while we load your content...
Please wait while we load your content...