Oxygen vacancy induced MnO2 catalysts for efficient toluene catalytic oxidation†
Abstract
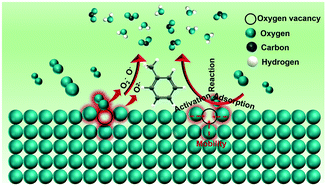
Oxygen vacancies play a vital role in catalytic oxidation, and engineering oxygen vacancies onto Mn oxide catalysts is still a great challenge. Herein, oxygen vacancies have been simply tuned on α-MnO2 by a facile redox method and used for catalytic toluene oxidation. The MnO2-1.8 catalyst exhibited superior catalytic activity (T90 = 238 °C, 1000 ppm and T90 = 170 °C, 10 000 ppm) for toluene deep oxidation, robust stability, and water resistance. It has the highest oxygen vacancy concentration due to highly proportioned exposed high-index {310} facets and interfaces between facets. The correlation between oxygen vacancies and the textural structure and activity was scientifically investigated by various characterization technologies and density functional theory (DFT) calculations. The oxygen vacancy weakens the Mn–O bond strength, increases the amount of surface adsorbed active oxygen species, and improves the mobility of lattice oxygen species, resulting in the significant promotion of the catalytic activity. This paper presents a novel and facile redox synthesis method for enriching the oxygen vacancy engineering strategy and provides a deep understanding for developing high-activity Mn-based catalysts for the VOC degradation.


 Please wait while we load your content...
Please wait while we load your content...