Highly selective electroreduction of nitrate to ammonia on a Ru-doped tetragonal Co2P monolayer with low-limiting overpotential†
Abstract
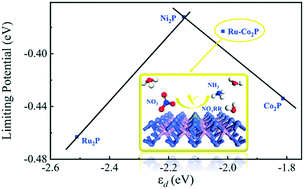
Selective electroreduction of nitrate (NO3−) into ammonia (NH3) can not only eliminate the water pollution crisis but also provide a sustainable way for low-temperature NH3 synthesis. However, today's development of the electrochemical NO3− reduction reaction (NO3RR) is hindered by the lack of eligible electrocatalysts and clear mechanism clarification. Here, the application potentials of the tetragonal transition-metal phosphide monolayers (M2P, M = Co, Ni, Ru and Pd) as NO3RR electrocatalysts for NH3 synthesis have been comprehensively and originally studied using density functional theory. Through comparing the relative energies of various possible products along the NO3RR process, a novel alternating pathway is proposed and it is beneficial to avoid the formation of N-containing by-products. Selecting the d-band center (εd) as an effective descriptor, a volcano-type plot of the NO3RR limiting potential is established. The Ni2P monolayer stands near the top of the volcano plot with the limiting potential of −0.37 V, but its selectivity can be greatly reduced when applying the electrode potential. Then, a Ru-doped Co2P monolayer is constructed to modulate the εd close to the value at the volcano top and its limiting potential is −0.38 V, exhibiting excellent activity. In addition, the Ru-doping process is thermodynamically favorable and the competing hydrogen evolution reaction is significantly suppressed even with the application of electrode potentials. This work not only provides an atomic understanding of the NO3RR process and but also offers a reliable descriptor to help design promising electrocatalysts.


 Please wait while we load your content...
Please wait while we load your content...