A computational study on the reduction of O2 to H2O2 using small polycyclic aromatic molecules†
Abstract
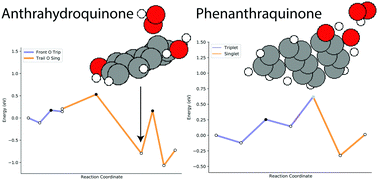
Hydrogen peroxide is an environmentally friendly oxidizing agent that is important in several industries. It is currently produced industrially via the anthrahydroquinone (AHQ) process where O2 reacts with a functionalised version of anthrahydroquinone to produce H2O2 and anthraquinone. In the previously published DFT pathway for this process the transition of the OOH˙ radical across the partially dehydrogenated AHQ catalyst was not explored. In this paper, we will use DFT to explore this step and show that there is a deep potential energy minimum that inhibits the OOH˙ from being fully reduced. We then examine other similar sized polycyclic molecules with two OH-groups on the same side that could serve as alternative catalysts without this issue. In this analysis, we identify phenanthraquinone as a possible alternative and present the pathway for this candidate to produce H2O2 as well as its regeneration with H2.


 Please wait while we load your content...
Please wait while we load your content...