Carbonyl umpolung as an organometallic reagent surrogate
Abstract
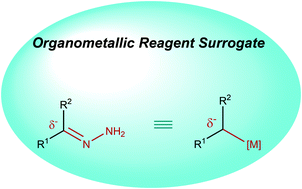
Construction of new carbon–carbon bonds is the cornerstone of organic chemistry. Organometallic reagents are amongst the most robust and versatile nucleophiles for this purpose. Polarization of the metal–carbon bonds in these reagents facilitates their reactions with a vast array of electrophiles to achieve chemical diversification. The dependence on stoichiometric quantities of metals and often organic halides as feedstock precursors, which in turn produces copious amounts of metal halide waste, is the key limitation of the classical organometallic reactions. Inspired by the classical Wolff–Kishner reduction converting carbonyl groups in aldehydes or ketones into methylene derivatives, our group has recently developed strategies to couple various alcohols, aldehydes, and ketones with a broad range of both hard and soft carbon electrophiles in the presence of catalytic amounts of transition metals, via the hydrazone derivatives: i.e., as organometallic reagent surrogates. This Tutorial Review describes the chronological development of this concept in our research group, detailing its creation in the context of a deoxygenation reaction and evolution to a more general carbon–carbon bond-forming strategy. The latter is demonstrated by the employment of carbonyl-derived alkyl carbanions in various transition-metal catalyzed chemical transformations, including 1,2-carbonyl/imine addition, conjugate addition, carboxylation, olefination, cross-coupling, allylation, alkylation and hydroalkylation.


 Please wait while we load your content...
Please wait while we load your content...