Experimental studies on combined production of CH4 and safe long-term storage of CO2 in the form of solid hydrate in sediment
Abstract
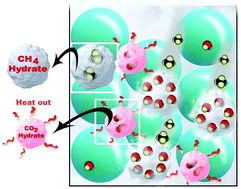
The production of the confirmed enormous resources of CH4 trapped in permafrost and deep ocean sediments in the form of hydrates has been hampered by the lack of an extraction procedure that is both effective and environmentally sensitive. This research explores experimentally the dynamic rate limiting steps in the dissociation of methane hydrates and the formation of CO2 hydrates in a sediment matrix. The use of CO2 injection and substitution for hydrate extraction takes advantage of novel thermodynamics and also provides a safe storage option for greenhouse gas. This experimental work incorporates a high-pressure facility dedicated for CH4 hydrates exchange with CO2 that replicates creation of natural gas hydrate from incoming gas below water in the pore space. The hydrate formation/exchange chamber follows the state-of-art hydrate science and is equipped with sensors distributed in several sections: the top section for gas release, a CH4 hydrate section, and a subsequent injection of CO2 from the bottom section, which also mimics hydrate dissociation towards incoming seawater through fracture systems connected from the seafloor. Four experimental conditions were examined. They comprise pure CO2 injection, and 10, 20, and 30 mole% N2 added to the CO2. We observed an increase in CH4 release from pure CO2 injection to 10 mole% N2 addition. A significant extra release of CH4 occurred by stepping up to 20 mole% N2 addition but no significant change was observed from 20 to 30 mole% N2 addition. Maximum conversion in this study is 34 mole% of CO2, and 2 mole% N2 taking the place of methane hydrate in large and small cavities. The results also show that effective substitution for hydrate production cannot rely on pure carbon dioxide injection.


 Please wait while we load your content...
Please wait while we load your content...