Enthalpy formation of fluorene: a challenging problem for theory or experiment?†
Abstract
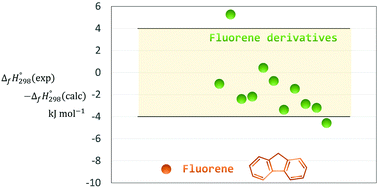
The large discrepancy between the experimental enthalpy of formation of fluorene and theoretical value calculated by the G3(MP2) method was revealed more than ten years ago. Three years later, a new experimental study of this compound was undertaken to ascertain whether there is any significant error in the thermochemical data. However, after this research, the agreement between theory and experiment was improved only slightly. In this work we decided to calculate the enthalpy of formation of fluorene using the high-level DLPNO-CCSD(T1)/CBS method which shows better results compared to Gn theories. To examine the accuracy of the available experimental data, the calculations were performed not only for fluorene but also for eleven fluorene derivatives. The discrepancy of about 9 kJ mol−1 between the experimental and theoretical enthalpies of formation of fluorene was confirmed by the present calculations, whereas good agreement was observed for the fluorene derivatives. It is highly unlikely that this discrepancy may disappear when using a higher-level theory. The possible reason for such inconsistency might be the experimental difficulty associated with the glass transition discovered in the stable crystalline state of fluorene. In this case, new experiments using the latest methods, such as differential scanning calorimetry combined with X-ray powder diffraction, are needed to gain deeper insight into the solid phase transformations of fluorene.


 Please wait while we load your content...
Please wait while we load your content...