Umbrella inversion of ammonia redux
Abstract
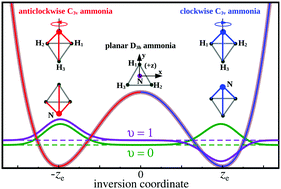
Umbrella inversion of ammonia is a prototypical example of large-amplitude vibrational motion, described with a symmetric double-well potential. The transition state of the latter corresponds to a planar (D3h) molecular geometry, whereas the two equilibrium configurations are equivalent (C3v) pyramidal structures, with the nitrogen atom being either ‘above’ or ‘below’ the plane of the hydrogen atoms. As commonly understood, inversion motion of ammonia corresponds to the coherent, anharmonic, vibrational motion of the molecule, which shuttles back and forth between the two potential wells; that is, oscillation of the nitrogen atom from one side of the H3 plane to the other, via coherent tunneling. However, this intuitively appealing view of umbrella inversion results from a reduced description of the dynamics, which includes only the inversion vibrational coordinate and fully neglects all the other molecular degrees of freedom. As such, this textbook picture of inversion motion ignores the fact that the two equilibrium structures of ammonia are superimposable, and can only be distinguished by labelling the identical hydrogen nuclei. A correct description of umbrella inversion, which incorporates nuclear permutations, requires the inclusion of other molecular modes. Indeed, it is well known that the quantum symmetrization postulate engenders entanglement between ammonia's nuclear-spin, inversion, and rotation. Using the explicit expressions of the corresponding zeroth-order eigenstates, we clearly show that the inversion density of any multilevel wavepacket of ammonia, including the case of perfectly aligned molecules, is symmetrically distributed between the two potential wells, at all times. This follows from a rigorous demonstration based on the evaluation of the expectation values of the inversion coordinate or equivalent projection operators. However, provided that these wavepackets involve inversion–rotation levels with opposite parity, the inversion density may exhibit dynamical spatial localization. In the latter case, the space-fixed inversion density or, equivalently, the expectation values of the projections of the inversion coordinate on the space-fixed axes, may oscillate between opposite directions in the space-fixed frame. Nevertheless, in all cases, localization of ammonia in a single potential well is impossible, even partially or transiently. This is equivalent to saying that the nitrogen atom has the same probability (one-half) to be on either side of the H3 plane, for any wavepacket of the molecule and at all times—a conclusion which is in perfect accord with the principle of the indistinguishability of identical particles (nuclei).


 Please wait while we load your content...
Please wait while we load your content...