The complex between molecular oxygen and an organic molecule: modeling optical transitions to the intermolecular charge-transfer state†
Abstract
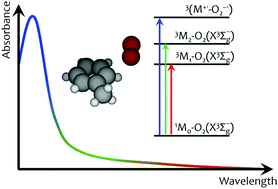
The collision complex between the ground electronic state of an organic molecule, M, and ground state oxygen, O2(X3Σg−), can absorb light to produce an intermolecular charge transfer (CT) state, often represented simply as the M radical cation, M+˙, paired with the superoxide radical anion, O2−˙. Aspects of this transition have been the subject of numerous studies for ∼70 years, many of which address fundamental concepts in chemistry and physics. We now examine the extent to which the combination of Molecular Dynamics simulations and electronic structure response methods can model transitions to the toluene–O2 CT state. To account for the experimental spectra, we consider (a) the distribution of toluene–O2 geometries that contribute to the transitions, (b) a quantitative description of intermolecular CT, and (c) oxygen-induced local transitions in toluene that complement the CT transitions, specifically transitions that populate toluene triplet states. We find that the latter oxygen-induced local transitions play a prominent role on the long wavelength side of the spectrum commonly attributed to the intermolecular CT transition. Our calculations provide a new perspective on the seminal discussion between R. S. Mulliken and D. F. Evans on the nature of O2-dependent transitions in organic molecules, and bode well for modeling transitions to excited states with CT character in noncovalent weakly-bonded molecular complexes.
- This article is part of the themed collection: 2021 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...