Preferential solvation of carbohydrates in water–trifluoroethanol mixtures: a solvent detected heteronuclear NMR approach†
Abstract
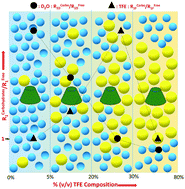
The present study aims to establish a simple approach involving multi-field multinuclear longitudinal relaxation (R1) analysis of the solvents to decipher solute–solvent interactions during the solvation of model carbohydrates in aqueous trifluoroethanol (TFE) co-solvent systems (TFE:D2O). The behavior of D2O and TFE is monitored around β-CD (β-cyclodextrin) and glucose through R1D (2H) and R1F (19F), respectively. Correlation times (τc) are estimated for D2O and TFE for various % (v/v) compositions of TFE:D2O mixtures. The differential trends of the R1 or τc ratio for D2O and TFE (in the presence and absence of carbohydrates) revealed that both β-CD and glucose undergo selective solvation by TFE in comparison to D2O. Owing to its encapsulation properties, β-CD exhibited a comparatively higher tendency to undergo solvation by TFE than glucose. The maximum transfer of solute bound water to bulk solvent appears in the 20–30% (v/v) TFE range. The current approach emerges as being straightforward in contrast to traditional methods that primarily focus on solute behavior to unravel the preferential solvation dynamics.


 Please wait while we load your content...
Please wait while we load your content...