Supramolecular architectures sustained by delocalised C–I⋯π(arene) interactions in molecular crystals and the propensity of their formation†
Abstract
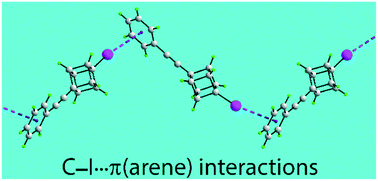
A systematic analysis of the Cambridge Crystallographic Database has been made for supramolecular architectures sustained by C–I⋯π(chelate ring) interactions where the iodide atom is directed towards the ring centroid, Cg, of the arene ring, i.e. with the angle subtended at the iodide atom, C–I⋯Cg (θ) being ≥160°. The majority of the 181 identified aggregates are one-dimensional chains of varying topology (100 examples) followed by zero-dimensional aggregates (71 examples) with only a small number of two-dimensional arrays (4 examples). The overall likelihood for the formation of these delocalised interactions is around 4%, a number that increases to around 15% when the angle θ restriction is relaxed to 90°. A comparison has been made with the formation of C–X⋯π(chelate ring) interactions for the lower X = bromide, chloride and fluoride congeners. This shows these interactions are more likely to form in the order X = I > Br > Cl ≫ F.
- This article is part of the themed collections: Introducing the CrystEngComm Advisory Board and their research and Supramolecular & Polymorphism

 Please wait while we load your content...
Please wait while we load your content...