Physical stability enhancement and antimicrobial properties of a sodium ionic cocrystal with theophylline†
Abstract
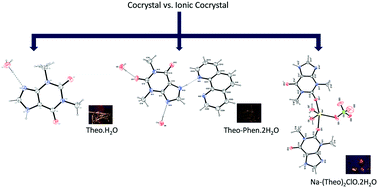
In the present study, we have described the synthesis and characterisation of the theophylline hydrate (Theo hydrate), cocrystal (Theo–Phen·2H2O) and hydrated sodium co-crystal of theophylline (Na–(Theo)2ClO·2H2O), where Theo = theophylline and Phen = 1,10-phenathroline. SC-XRD characterisation revealed strong interactions such as N⋯H⋯O, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O⋯H and C–H⋯π within the crystal structures. Studies revealed that Na–(Theo)2ClO·2H2O presented higher thermal stability (in the solid form) and improved solubility (when in contact with a polar solvent). The detailed analyses of Hirshfeld surface and fingerprint plots provided insight into the nature of non-covalent interactions in the compounds. In the case of Na–(Theo)2ClO·2H2O, solubility measurements indicated that Theo's solubility in water increases by a magnitude of four with respect to pure Theo. Biological studies showed that the high solubility of Theo as well as ClO4− is responsible for the distinct capabilities to inhibit the growth of different microorganisms, including Gram-positive (Staphylococcus epidermidis) and Gram-negative (Acinetobacter baumannii, Escherichia coli and Klebsiella pneumoniae) bacteria and fungi (Candida albicans and Candida tropicalis). Our results pointed out that cocrystals containing theophylline–phenanthroline can be explored to get promising molecules to be used to combat infections caused by clinically relevant (multi)drug-resistant bacteria and fungi.
O⋯H and C–H⋯π within the crystal structures. Studies revealed that Na–(Theo)2ClO·2H2O presented higher thermal stability (in the solid form) and improved solubility (when in contact with a polar solvent). The detailed analyses of Hirshfeld surface and fingerprint plots provided insight into the nature of non-covalent interactions in the compounds. In the case of Na–(Theo)2ClO·2H2O, solubility measurements indicated that Theo's solubility in water increases by a magnitude of four with respect to pure Theo. Biological studies showed that the high solubility of Theo as well as ClO4− is responsible for the distinct capabilities to inhibit the growth of different microorganisms, including Gram-positive (Staphylococcus epidermidis) and Gram-negative (Acinetobacter baumannii, Escherichia coli and Klebsiella pneumoniae) bacteria and fungi (Candida albicans and Candida tropicalis). Our results pointed out that cocrystals containing theophylline–phenanthroline can be explored to get promising molecules to be used to combat infections caused by clinically relevant (multi)drug-resistant bacteria and fungi.
- This article is part of the themed collection: Crystal Growth


 Please wait while we load your content...
Please wait while we load your content...