Carbon monoxide bond cleavage mediated by an intramolecular frustrated Lewis pair: access to new B/N heterocycles via selective incorporation of single carbon atoms†
Abstract
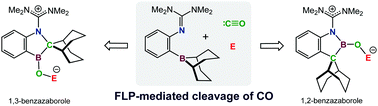
Utilizing an intramolecular frustrated Lewis pair (FLP) decorated with a strongly donating guanidino moiety enabled the formation of a thermally remarkably stable FLP-CO adduct, which at 120 °C underwent CO migration to form an acyl borane. Both compounds underwent rapid CO cleavage in the presence of strong electrophiles leading to the selective formation of a range of new 1,2- and 1,3-benzazaboroles in good yields under mild conditions.
- This article is part of the themed collection: Boron Chemistry in the 21st Century: From Synthetic Curiosities to Functional Molecules


 Please wait while we load your content...
Please wait while we load your content...