Atomic-layered Pt clusters on S-vacancy rich MoS2−x with high electrocatalytic hydrogen evolution†
Abstract
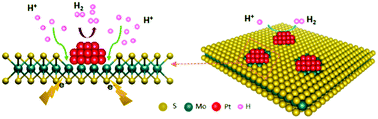
Developing suitable supports to maximize the atomic utilization efficiency of platinum group metals is of great significance to hydrogen evolution from water splitting. Herein, we report a fully exposed Pt cluster supported on an S-vacancy rich MoS2−x support (Pt/Sv-MoS2−x) by a facile impregnation method. Pt/Sv-MoS2−x exhibits an outstanding electrochemical HER performance with a low overpotential of 26.6 mV at a current density of 10 mA cm−2, a small Tafel slope of 34.8 mV dec−1 and good durability. Most importantly, the mass activity of Pt is an order of magnitude more active than that of commercial Pt/C at an overpotential of 0.08 V. We attribute this exceptional HER catalytic performance to the fact that platinum and Sv-MoS2−x act in synergy to accelerate the reaction kinetics.


 Please wait while we load your content...
Please wait while we load your content...