Catalyst-controlled site-selective N–H and C3-arylation of carbazole via carbene transfer reactions†
Abstract
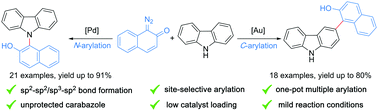
A site-selective direct arylation reaction of carbazole and other N-heterocycles with diazo-naphthalen-2(1H)-ones has been developed. While Au(I)-NHC catalysts lead to selective C3-arylation, palladium acetate allows for selective N–H arylation, displaying complete site-selectivity each. To show the applicability of these arylation reactions, one-pot, two-fold diarylation reactions of carbazole were demonstrated.
- This article is part of the themed collection: Functionalization of unreactive C-H bonds


 Please wait while we load your content...
Please wait while we load your content...