Diketopyrrolopyrrole based donor–acceptor π-conjugated copolymers with near-infrared absorption for 532 and 1064 nm nonlinear optical materials†
Abstract
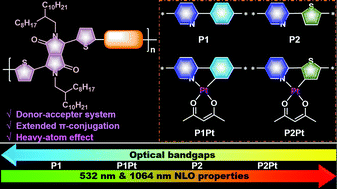
Organic and organometallic π-conjugated molecules with inherent nonlinear optical (NLO) responses have attracted great interest due to their controllable properties via rational structure modifications. However, most of the state-of-the-art materials such as porphyrins, phthalocyanines, and diacetylenes exhibit NLO responses at visible-light regions. Recently, diketopyrrolopyrrole (DPP) based donor–acceptor (D–A) type π-conjugated copolymers have been found to exhibit NLO responses which can reach up to 1064 nm. However, the design rationality of the structure toward NLO properties is not well understood. Herein, four alternating DPP-based copolymers are constructed by using the electron donors of 2-phenylpyridine for P1, 2-thienylpyridine for P2 and their Pt(II)-incorporated moieties for P1Pt and P2Pt. All copolymers display intense intramolecular charge transfer (ICT) processes, and the ICT absorption can be extended to the near-infrared (NIR) region due to the strong D–A interaction of the π-conjugated backbone. The variation tendency of optical bandgaps estimated from the onset of ICT absorption is P1 (1.60 eV) > P1Pt (1.56 eV) > P2 (1.47 eV) > P2Pt (1.44 eV). Open- and closed-aperture Z-scan experiments disclosed that all copolymers exhibit broadband (532 and 1064 nm) NLO properties due to the synergistic effect of excited state absorption and the ICT process upon excitation of 8 ns as well as 25 ps laser pulses. Pt(II)-incorporation can promote or switch the NLO properties due to the efficient triplet-state absorption introduced by the heavy-atom effect. The improvement of the NLO properties is of the order P1 < P1Pt < P2 < P2Pt, which is consistent with the decreasing trend of the copolymer bandgaps. These results will provide guidelines for the rational design of materials toward broadband NLO responses and advanced applications.


 Please wait while we load your content...
Please wait while we load your content...