A novel donor moiety 9,9,9′9′-tetramethyl-9,9′10,10′-tetrahydro-2,10′-biacridine via one-pot C–H arylation for TADF emitters and their application in highly efficient solution-processable OLEDs†
Abstract
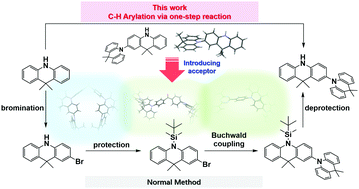
9,10-Dihydroacridine is a very popular donor unit for the construction of thermally activated delayed fluorescence (TADF) molecules due to its good electron-donating properties and potential for chemical modifications. However, the functionalization of the 2/7-positions of the acridine unit has rarely been explored. In this contribution, the novel donor moiety 9,9,9′9′-tetramethyl-9,9′10,10′-tetrahydro-2,10′-biacridine (BDMAc) was prepared via a one-pot C–H arylation procedure. Different acceptor fragments, diphenylsulfone (DPS), benzophenone (BPO) and 1,3,5-triazine (TRZ), were then introduced to synthesize three donor–acceptor molecules: DPS-BDMAc, BPO-BDMAc and TRZ-BDMAc. Single crystal X-ray diffraction and theoretical calculations show that these molecules possess twisted molecular geometries and small energy gaps between the singlet and triplet excited states. All molecules exhibit TADF properties with emission peaks between 480–600 nm in both solution and solid states, along with a high emission efficiency of 69–89% in the solid state. Solution-processed organic light-emitting diodes (OLEDs) based on these molecules were prepared with 25 wt% dopant concentration. Although the devices showed efficiency roll-off, a promising maximum EQE of ∼23% and a luminance of 7173 cd m−2 were obtained for the BPO-BDMAc-based device. This research demonstrates the potential of this novel acridine group for designing TADF molecules for efficient solution-processable OLEDs.


 Please wait while we load your content...
Please wait while we load your content...