Enzyme-like MOFs: synthetic molecular receptors with high binding capacity and their application in selective photocatalysis†
Abstract
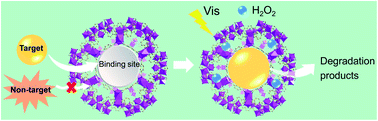
Metal–organic frameworks (MOFs) are promising materials in the separation and catalysis fields. However, traditional MOFs usually show limited defects and poor binding selectivity. Designing a versatile strategy to address these issues is therefore a great challenge. In this work, by integrating molecular imprinting and MOF fabrication, imprinted MOFs with specific recognition defects were generated. An adsorption isotherm study indicated that the imprinted UiO-66_NH2 had a binding capacity of 295.2 mg g−1 towards the ketoprofen template, which is much higher than that of traditional molecularly imprinted polymers (MIPs). The universality of this synthetic strategy was verified by creating specific cavities in different types of imprinted MOFs (e.g., MIL-101_NH2 and UiO-66_NH2). Remarkably, the imprinted MOFs (represented by imprinted MIL-101_NH2) exhibited significantly enhanced photocatalytic activity for target sulfadimidine degradation. In comparison with the non-imprinted MIL-101_NH2 system, the imprinted MIL-101_NH2 showed much higher photocatalytic activity (∼64%) and larger photocatalytic selectivity (4.74). We believe that this versatile strategy provides new insights into the design of enzyme-like MOFs with many defects and high binding selectivity.


 Please wait while we load your content...
Please wait while we load your content...