Integrated selective nitrite reduction to ammonia with tetrahydroisoquinoline semi-dehydrogenation over a vacancy-rich Ni bifunctional electrode†
Abstract
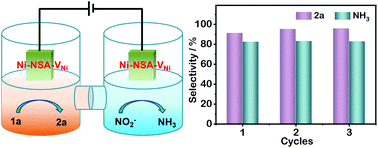
The development of efficient electrocatalysts for nitrite reduction to ammonia, especially integrated with a value-added anodic reaction, is important. Herein, Ni nanosheet arrays with Ni vacancies (Ni-NSA-VNi) were demonstrated to exhibit outstanding electrocatalytic performances toward selective nitrite reduction to ammonia (faradaic efficiency: 88.9%; selectivity: 77.2%) and semi-dehydrogenation of tetrahydroisoquinolines (faradaic efficiency: 95.5%; selectivity: 98.0%). The origin and quantitative analyses of ammonia were performed by 15N isotope labeling and 1H NMR experiments. The decrease in electronic cloud density induced by the Ni vacancies was found to improve the NO2− adsorption and NH3 desorption, leading to high nitrite-to-ammonia performance. In situ Raman results revealed the formation of NiII/NiIII active species for anodic semi-dehydrogenation of tetrahydroisoquinolines on Ni-NSA-VNi. Importantly, a Ni-NSA-VNi‖Ni-NSA-VNi bifunctional two-electrode electrolyzer was constructed to simultaneously produce ammonia and dihydroisoquinoline with robust stability and high selectivity.


 Please wait while we load your content...
Please wait while we load your content...