A high performance direct borohydride fuel cell using bipolar interfaces and noble metal-free Ni-based anodes†
Abstract
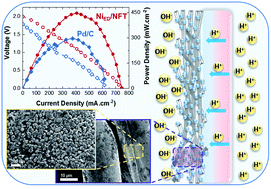
Due to its unmatched theoretical voltage of 2.18 V, a direct alkaline fuel cell using sodium borohydride solution at the anode and hydrogen peroxide at the cathode represents a promising power source for high energy density applications. However, its development faces several challenges. Here we demonstrate a BH4−/H2O2 direct borohydride fuel cell (DBFC) with a platinum group metal (PGM)-free anode, which delivers an unprecedented combination of 2.0 V open-circuit voltage and a peak power density of 446 mW cm−2. This exceptionally high cell voltage is enabled by combining a pH-gradient-enabled microscale bipolar interface (PMBI), a Ni anode obtained by electrodeposition of Ni nanoparticles on a chemically etched Ni felt (eNFT), and a specially designed simple but efficient coating procedure to deposit anion-exchange ionomers on the anode surface. The PMBI efficiently separates the drastically disparate pH of the anolyte and the catholyte, the NiED/eNFT anode provides a high surface area for efficient electrocatalysis and open porosity for fast mass-transport, while the coating procedure allows preserving Ni in the metallic state, the latter being a prerequisite for high anode performance. This work details how such fully nickel-based anodes are obtained and demonstrates why their BOR activity and stability outperform those of PGM-based anodes.


 Please wait while we load your content...
Please wait while we load your content...